About the Project

THE CANDIDATE STUDY
Study Title: A study to develop an algorithm utilising artificial intelligence to predict the likelihood of patients completing the dose limiting toxicity assessment period of early phase cancer clinical trials. A study limited to working with retrospective data.
Study funder: The project is being funded through the UPSMART programme.
UK Study Lead: Dr Louise Carter
UK Study Contact: the-christie.CANDIDATE@nhs.net
Project Background
Clinical trials for new cancer treatments have strict rules on which patients can participate. Patients are usually required to undergo additional testing procedures to assess if they meet these study requirements before they begin treatment. Despite these additional tests it is still common for patients to be found to be unsuitable for the trial or to stop treatment early, for a reason other than side effects from the drugs. Patients that stop treatment within the first 28 days of a trial are unlikely to receive any benefit whilst still being at risk from side effects from the medication. Currently, the clinical team rely on their expertise and experience to decide whether patients are likely to be suitable for trials and will remain on a trial for longer than 28 days.
If we could improve the assessment of patients’ suitability for trials it could reduce unnecessary distress and potential harm to patients. It could also reduce delays in the testing of new drugs, as patients are more likely to remain on study long enough to obtain the information about the possible side effects of new treatments needed to complete the study.
In this study we aim to assess if we can use artificial intelligence to analyse sections of patients’ hospital notes to develop a tool (algorithm) which can predict if patients are likely to be suitable for clinical trials and remain on them for longer than 28 days. The study will be conducted by researchers at the University of Manchester, The Christie NHS Foundation Trust, Manchester, UK, and The Centre Léon Bérard, Lyon, France.
The following relates specifically to activity in the UK at The Christie NHS Foundation Trust.
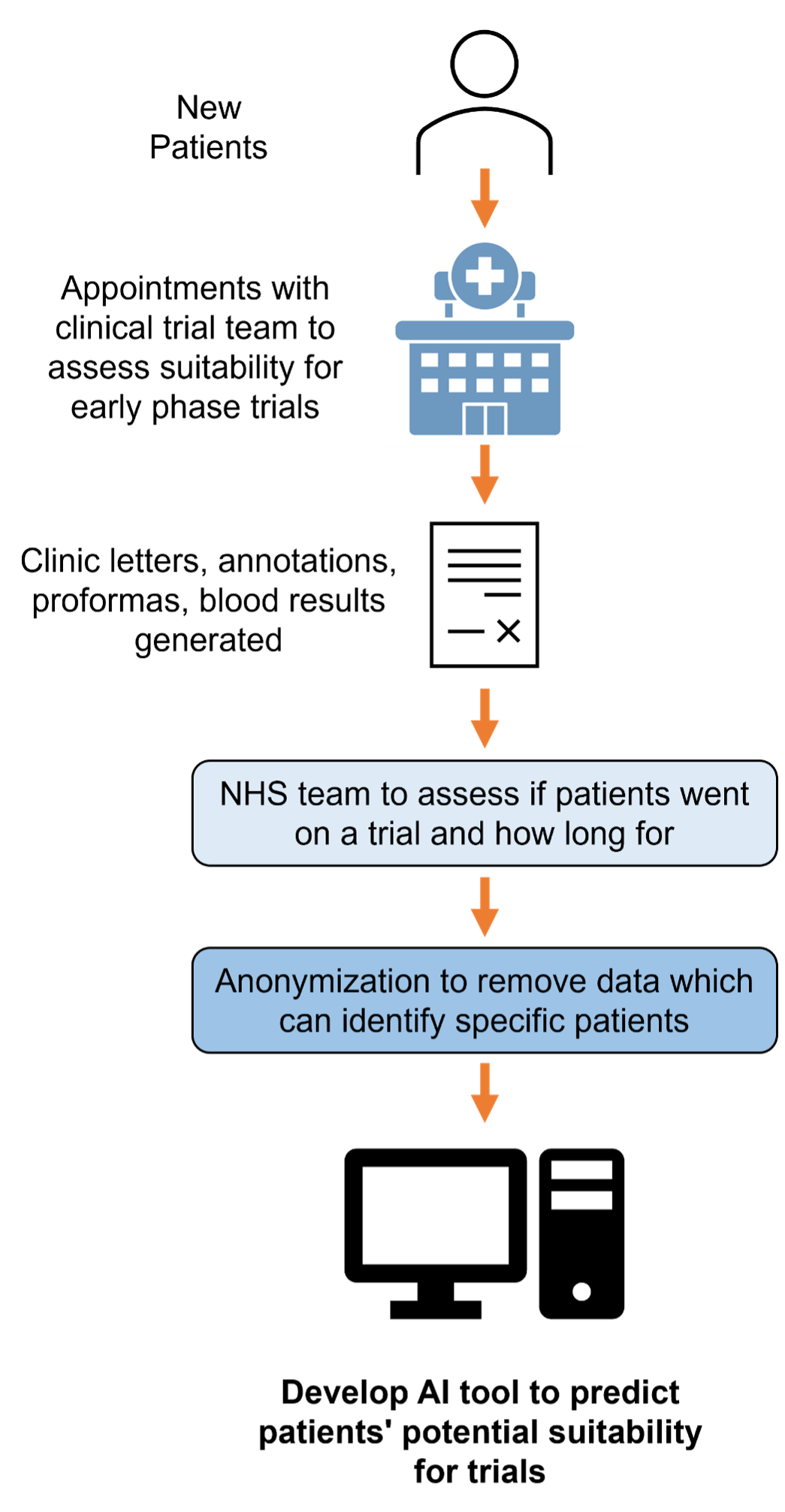
How is patients’ data anonymised and stored?
Before patients’ data is used in the research it will be anonymised so that elements that could identify specific patients such as names will be removed. To do this the Christie NHS Foundation Trust will process the data using a computer programme designed to remove patient identifiable information. Small amounts of identifiable data may still remain after this.
The anonymised data is collected and kept on secure NHS computers. Access to it will be controlled and monitored by The Christie NHS Foundation Trust. The artificial intelligence tool does not keep or copy the data that is has analysed. Researchers will not be able to identify any specific patients or their data from the artificial intelligence tool created by the research.
This is an Opt-Out study
This means if you had a New Patient clinic appointment between 1st January 2017 and 31st December 2023 with the Experimental Cancer Medicine team at the Christie, data relating to your appointment and subsequent enrolment into an early phase clinical trial will be included in the study unless you tell us not to or have used the National Data Opt-Out
What happens if my data is used?
- If you are a current patient, nothing about your cancer treatment will change.
- The study will use patients’ data that is collected during new patient appointments with the clinical trial team including letters, blood results and proformas. This data includes identifiable information about specific patients such as their name. The clinical team will review patients’ histories to confirm if patients were recruited to a trial and if so were they then found to be suitable for the trial and the length of time on they were remained on it. The data will then be anonymised (identifiable information such as names removed) before being analysed in the research. Only a patients clinical team will have seen the data before it is anonymised.
- All of your information will be stored safely, securely and confidentially on a computer system at The Christie NHS Foundation Trust, only accessible to specifically approved staff for the purposes of this project.
- At the end of the study, we will securely maintain the database for a period of 5 years before it is securely destroyed.
How do I opt out?
- If you do not want your information to be used in this study, you must tell us. This will not change your cancer treatment.
- This is called opting out and you can do it without giving a reason.
- It will always be your choice to opt out.
If you decide you do not want your data to be used in the study, you can either use the national NHS data opt out or specifically opt out of this study by contacting the study team on: the-christie.CANDIDATE@nhs.net
If you prefer to call the Experimental Cancer Medicine Team at the following telephone number: 0161 918 7672. The team will document your request to opt-out of the CANDIDATE study and inform the study lead – Dr. Carter.
The national data opt-out https://digital.nhs.uk/services/national-data-opt-out is a service that allows patients to opt out of their confidential patient information being used for research and planning.
You can view or change your national data opt-out choice at any time by:
- using the online service at nhs.uk/your-nhs-data-matters or
- by clicking on “Your Health” in the NHS App, and selecting “Choose if data from your health records is shared for research and planning”.
You can find out more about how we use your information in Manchester:
- By reading the Manchester Cancer Research Centre patient and research data statement: https://www.mcrc.manchester.ac.uk/wp-content/uploads/2021/09/MCRC-Patient-Data-Statement-2021-09-01.pdf
- The “How the NHS Uses Data for Research” section of the Christie NHS Foundation Trust’s Privacy Notice: https://www.christie.nhs.uk/about-us/the-christie-nhs-foundation-trust-privacy-notice
- By sending an email to the-christie.CANDIDATE@nhs.net
Acknowledgements:
This project and accompanying information poster were developed in collaboration with patient contributors.
UK Ethics Approvals
The application was reviewed by the Confidentiality Advisory Group (CAG). CAG is an independent group of lay people and professionals which provides expert advice on the use of confidential patient information without consent. CAG recommended that our application should be supported and the Secretary of State for Health/Decision Maker within the Health Research Authority approved this.
Version 1.1, 12/03/2025
IRAS ID: 323831
Study title: A study to develop an algorithm utilising artificial intelligence to predict the likelihood of patients completing the dose limiting toxicity assessment period of early phase cancer clinical trials. A study limited to working with retrospective data.
Short Study title: CANDIDATE
QUICK LINKS
CONTACT

